Our Approach
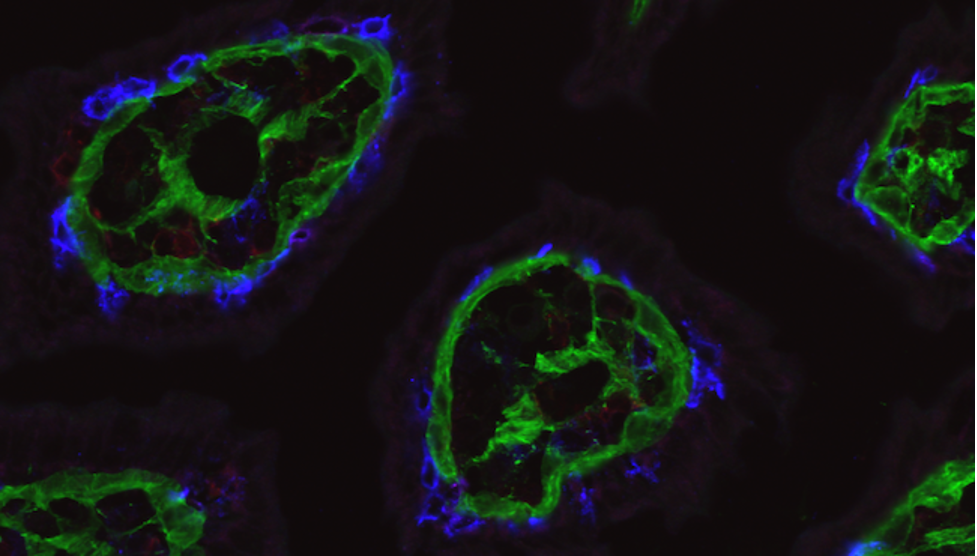
Researchers at La Jolla Institute for Immunology (LJI) are working to prevent the symptoms of IBD before inflammation escalates and damages the GI tract. Professor Mitchell Kronenberg, Ph.D., LJI’s Chief Scientific Officer, studies molecular signals used by inflammatory cells in IBD. Those analyses show that a membrane protein called HVEM, which resides on the surface of many cells, can bind two very different proteins, an inhibitory factor called BTLA and a pro-inflammatory cytokine called LIGHT. When HVEM binds LIGHT, the immune response increases and inflammation ramps up, but when it binds BTLA, the immune response decreases. His group is now seeking ways to flip the switch to favor signaling through BTLA to reduce inflammation in the intestine and they are testing forms of HVEM with selective binding only to BTLA.
Dr. Kronenberg’s laboratory has also shed light on what keeps specialized T cells in the gut in the first place. In 2022, the researchers reported that the barrier cells that line the intestine use HVEM as a way to communicate with patrolling T cells. This communication is essential for getting T cells to fight pathogens in the gut. For people with IBD, scientists may want to dampen these HVEM messages to reduce inflammation.
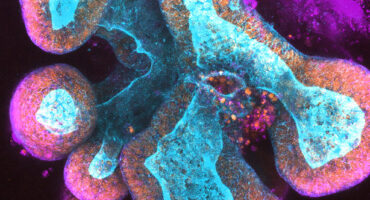
LJI Professor Hilde Cheroutre, Ph.D., addresses diseases of the gastrointestinal tract by studying T lymphocyte activity at the body’s “interfaces” with the outside world, such as skin, mouth and the largest surface of all, the intestines. Her lab investigates why T cells sometimes respond in such an inflammatory fashion in intestinal mucosa in the context of IBD.
Together, Drs. Kronenberg and Cheroutre are revealing the workings of T cells in the intestine, both in protecting the organ from infection and in causing IBD.
Learn more: