Our Approach
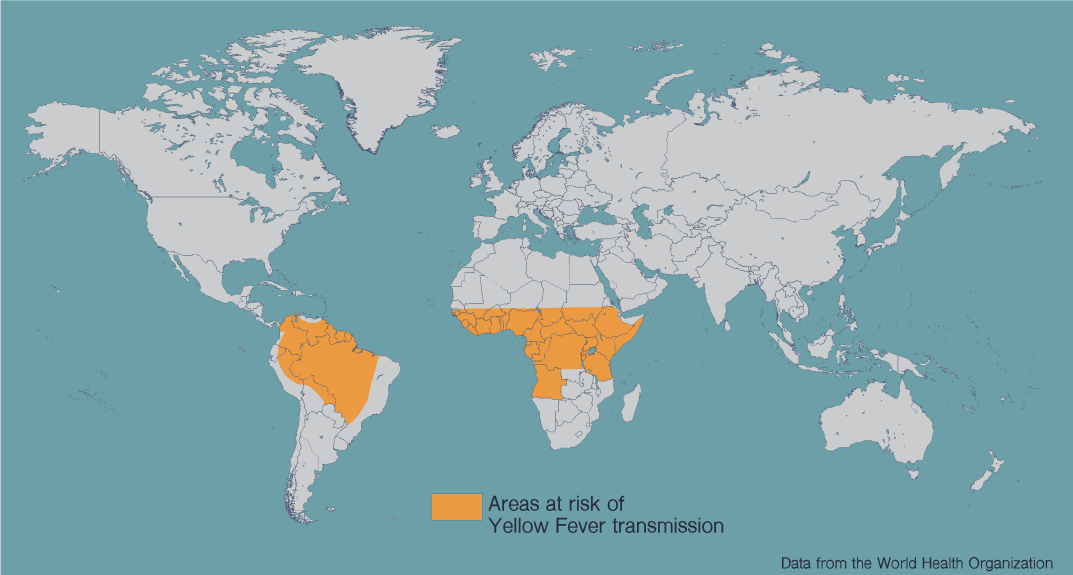
Thus, the YF vaccine is considered a benchmark, one of vaccine biology’s great success stories. As such, LJI vaccine biologist Alessandro Sette, Ph.D., thinks we must learn how it works if we are to design effective vaccines against related pathogens. To do so, his laboratory is now partnering with clinicians to collect samples from people who have been or will be vaccinated against YF.
Supported by a NIAID’s Human Immune Profiling Consortium, they will compare the repertoire of T cells found in their bloodstream to the immune counterparts in the blood of people vaccinated with an experimental vaccine to dengue, which like yellow fever (and West Nile) is what is called flavivirus. Since YF is one of the most successful vaccines to date it serves as a proxy for what protective immunity might look like against dengue, for which there is no vaccine.