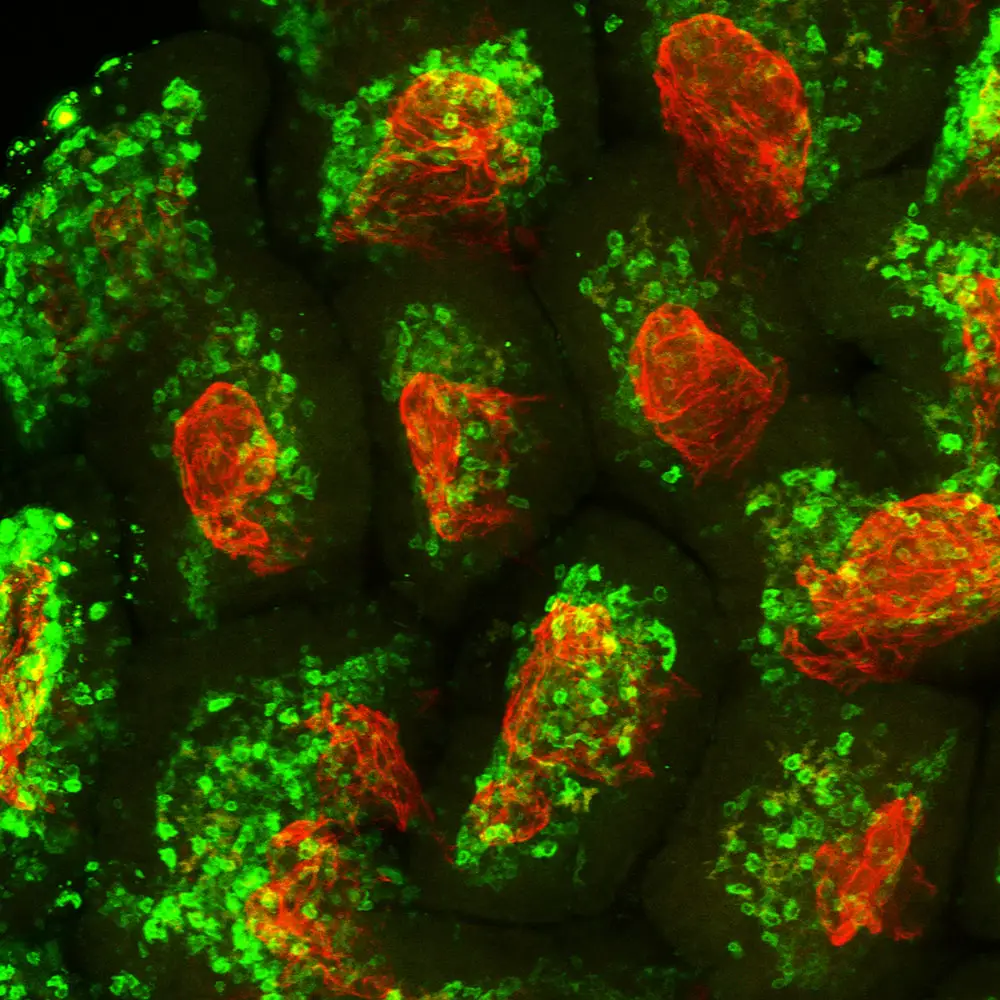
CMV
Cytomegalovirus (CMV) is a herpesvirus similar to “mono” (Epstein-Barr) or chicken pox (Varicella zoster) viruses. Half of all Americans are unknowingly infected with CMV, as the virus is adept at evading immune system defenses.
In most people, latent CMV infection is benign and goes unnoticed. The fever, pneumonia or ulcers that mark active infection emerge only when a person becomes immunocompromised, for example, when transplant recipients are treated with immunosuppressants.
But many are still vulnerable to the devastating effects of CMV. CMV infection is the number one infectious cause of congenital birth defects worldwide. In the United States, 1 in 700–800 babies is infected in utero and born with the infection. Anti-viral drugs can treat these infections, but these infants remain at risk for hearing loss, brain damage and even death.
CMV flare ups are also a major problem. Like chicken pox, which can reemerge 50 years later as shingles, latent CMV infection can flare up if the immune system becomes weakened, as in the elderly, possibly contributing auto-immune vascular disease or some cancers.
There is no FDA-approved CMV vaccine.
Our Approach
LJI scientists are working to define the molecular underpinnings of CMV infection and design effective vaccine strategies.
As a rapid response to infection, many cells display so-called “death receptors” that trigger cell death when activated by a cytokine called TRAIL. This “cell suicide” response helps clear out cells that could spread the virus. Several years ago, LJI Associate Professor Chris Benedict, Ph.D., discovered that CMV eludes that immune response by producing a protein called UL141, which jams those TRAIL signals. This uncanny ability to sabotage TRAIL signals explained how CMV-infected cells could survive and allow the infection to persist.
Dr. Benedict has identified a group of innate immune cells called ILC1s as the major cell type responsive to TRAIL signals. These cells are closely related to other innate immune cells named natural killer (NK) cells, which also mount an immediate response to some viral infections. Using mouse models, Dr. Benedict has discovered that it is ILC1s, not NK cells, that actually “contain” the CMV via unique mechanisms. Since ILC1s also exist in humans, this work furthers our understanding of how one might recruit these cells in an CMV vaccine.
In fact, in a 2024 study, Dr. Benedict and his colleagues uncovered another weapon against CMV. The researchers found that a special population of “late-rising” CD4+ “helper” T cells may help a person fight CMV and reduce the chances of the virus spreading from person to person.
Dr. Benedict has also identified unique CMV protein structures, or “epitopes,” recognized by T cells in both mice and man. One of these epitopes induces a unique ‘late rising’ T cell type that completely resolves CMV replication that can persist for many months. His lab found that vaccination with this and other epitopes elicits T cells that mediate dramatic protection in mice, indicating that it should also be possible to implement similar preventative approaches in humans.
LJI scientists launch new CMV studies
LJI scientists are now building on this CMV research and leading efforts to boost CMV vaccine research across the United States. LJI Professor, President, and CEO Erica Ollmann Saphire, Ph.D., MBA, recently received ARPA-H funding to launch a project called America’s SHIELD: Strategic Herpesvirus Immune Evasion and Latency Defense.
Under Dr. Saphire’s leadership, this national team to develop life-saving vaccines against CMV and a broad array of herpesviruses that infect the majority of Americans and can cause cancer, autoimmune disease, and birth defects. The award is part of ARPA-H’s Antigens Predicted for Broad Viral Efficacy through Computational Experimentation (APECx) program.
In a recent investigation, Drs. Benedict and Saphire worked with collaborators at the University of Pittsburgh School of Medicine to uncover a previously unappreciated mechanism by which CMV enters cells that line the blood vessels and contributes to vascular disease. By understanding this key step in CMV infection, scientists are closer to developing vaccines and therapies to prevent severe disease.
Learn more:
Related News
- Behind the Science, Blog
- Research News
- Institute News
Research Projects
We have recently discovered a novel connection between the TNF-related apoptosis inducing ligand (TRAIL) cytokine system and CMV. TRAIL is a TNF-family cytokine that can bind to several receptors (TRAIL [...]
HCMV-specific CD8 T cells protect bone marrow transplant patients when used in cellular immunotherapy, and their long-term maintenance is enhanced by CD4 T cells. Importantly, a robust HCMV-specific CD4 T [...]
Virulent isolates of CMV encode the ul144 orf, and we identified UL144 as an orthologue of the herpesvirus entry mediator (HVEM), a member of the TNF receptor superfamily. When the [...]