Lassa Fever
Lassa fever is caused by an arenavirus endemic to West Africa and transmitted to humans through exposure to infected rodent waste or via human-to-human contact. Many infected individuals exhibit mild malaise or a short fever. Unfortunately, a small percentage of those infected can develop fatal hemorrhagic fever, as grimly illustrated after a 2019 outbreak in Nigeria that killed nearly a quarter of the more than 600 individuals infected.
Globally, the CDC estimates that of the 100,000–300,000 individuals who will contract Lassa yearly, nearly 5,000 will die. Early treatment with the anti-viral ribavirin provides some relief, but there is no vaccine available to prevent this disease, despite its lethality.
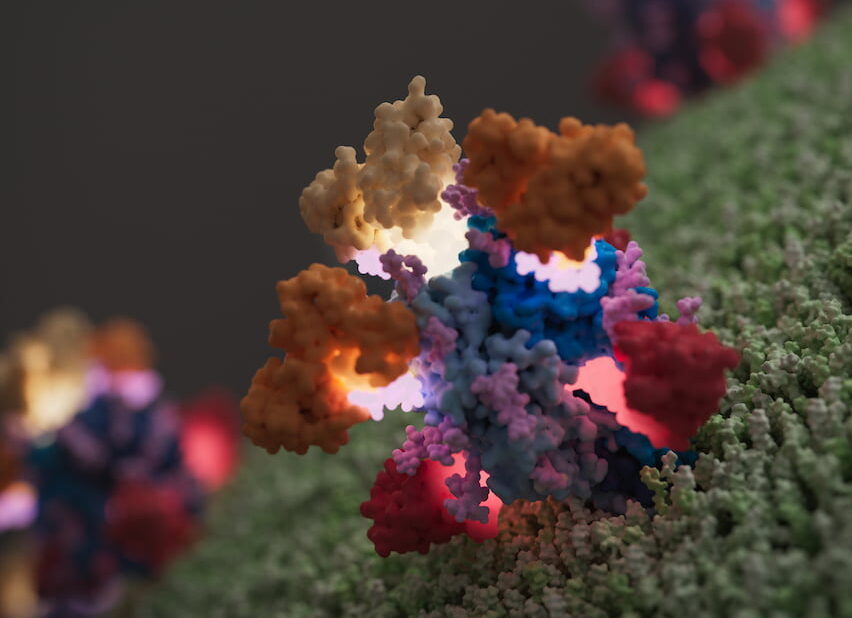
To meet this challenge, LJI scientists are conducting structural studies of a critical Lassa virus protein using next-gen imaging and microscopy. These studies lay the groundwork for an anti-Lassa vaccine and could provide the molecular insights required to engineer protective monoclonal antibodies as therapy for patients already infected.
Our Approach
LJI structural biologist Erica Ollmann Saphire, Ph.D., is a world leader in studies of viral proteins that could be targeted to block spread of Lassa fever or Ebola. In 2017, she was the first to report the crystal structure of a Lassa virus glycoprotein in direct contact with an antibody found in the blood of a Lassa survivor. That image was the first snapshot of how an antibody capable of blocking infection directly interferes with an essential viral protein, and it provided a template for vaccine design.
This work has challenged the assumptions about Lassa. Prior to Dr. Saphire’s discoveries, vaccine researchers had been skeptical that antibody-mediated immunity could effectively combat Lassa. Researchers had found that vaccine candidates did not seem to stimulate the immune system to potent neutralizing antibodies. Studies had also shown that few Lassa survivors had high titers of anti-Lassa antibodies in their bloodstream. Further complicating vaccine development efforts, Lassa is caused by four similar but molecularly-distinct variants or strains, and their prevalence differs from country to country.
To address these issues, in 2019 Dr. Saphire expanded structural analysis of Lassa’s outer protein spikes, called glycoproteins, this time scrutinizing how human antibodies target the glycoprotein with low, moderate and high affinity. This research compared molecular components of the target glycoprotein that either hindered or promoted high affinity interactions against all four strains.
Thanks to a collaboration with Scripps Research, the Dr. Saphire Lab has also uncovered a potential drug target for Lassa virus. In 2022, the researchers showed how a critical Lassa virus protein, called polymerase, drives infection by harnessing a cellular protein in human hosts. Their work suggests future therapies could target this interaction to treat patients. The next step is to start testing this theory in animal models.
The Saphire Lab also recently published research showing how an antibody cocktail with a trio of antibodies works to neutralize the virus. These antibodies may prove valuable in upcoming clinical trials for Lassa therapies, and the LJI team plans to use their new map of the Lassa virus surface glycoprotein to design a much-needed vaccine. This work was led by LJI Instructor Kathryn Hastie, Ph.D., who also serves as Director of LJI’s Center for Antibody Discovery.
These findings have fueled a global effort to engineer proteins that could make up an effective “pan-Lassa” vaccine. This ongoing research is a focus of international efforts underway by the Coalition for Epidemic Preparedness Innovations (CEPI), which has made Lassa fever a priority disease. In 2023, LJI Professor Alessandro Sette, Dr.Biol.Sci., and LJI Research Assistant Professor Alba Grifoni, Ph.D., received CEPI funding to collaborate with researchers across the country to study how to stop viruses from the Paramyxovirus and Arenaviruses families, which include Nipah virus and Lassa virus, respectively.
Learn more:
Related News
- Research News
- Institute News
- Research News