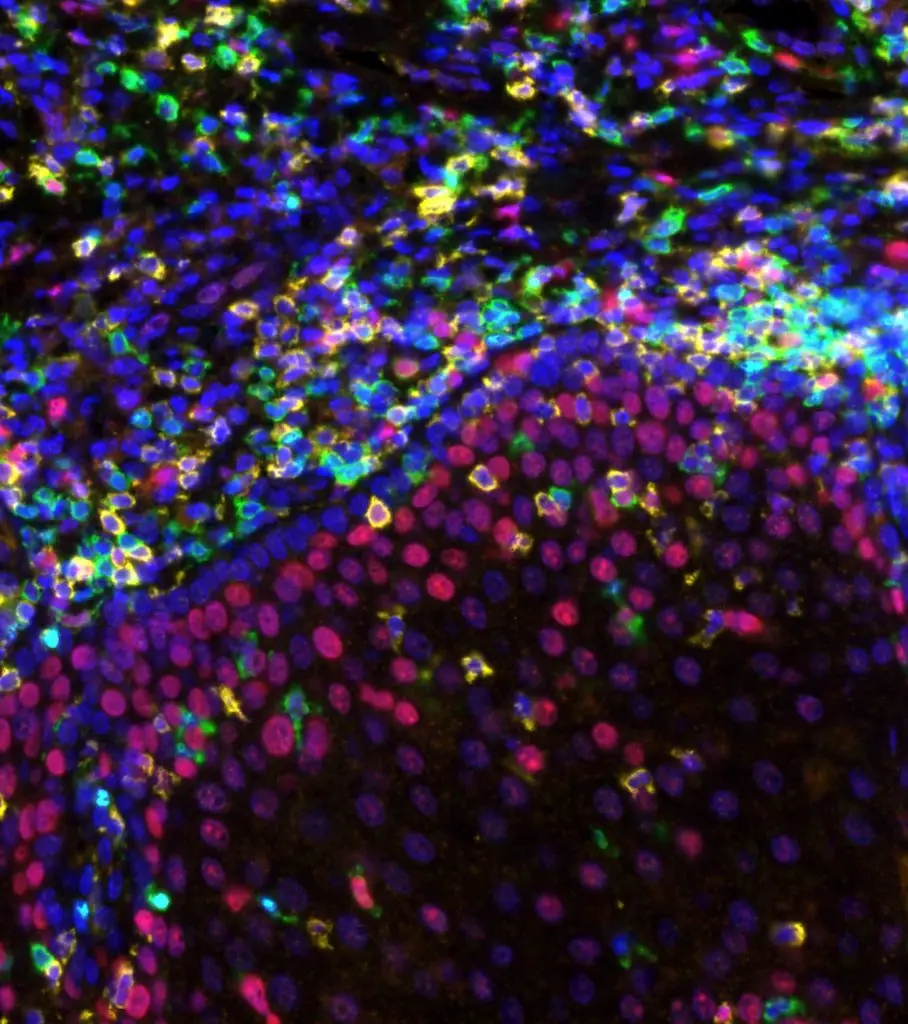
Cancer is a disease of genes, thought to arise in a single cell when those controlling growth and survival progressively sustain function-altering mutations during the process of tumor development. The immune system contains a specialized subset of lymphocytes called T cells, which possess the ability to recognize and kill our own cells when these are found to express foreign genes that signal the presence of a pathogenic virus or bacteria within. Mounting evidence has revealed that this same process which keeps us safe from harmful infections can be used to eradicate cancer by directing T cells to attack the mutations that are exclusively expressed in the tumor cells. Tumor-specific mutations that are recognized by an individual patient’s T cells are called neoantigens, and can form the basis for personalized cancer immunotherapy. Neoantigens can now be readily identified through genomic sequencing and immune responses to them can be generated through vaccination and measured at the level of single cells. Thus, all the tools needed to evaluate and optimize personalized immunotherapeutic approaches to cancer treatment are in place.
My laboratory’s current research is focused on achieving a mechanistic understanding of the generation and regulation of T cell responses in the context of in vivo infection and tumor development. Our work is focused on three main areas. One of these concerns the role of CD4+ ‘helper’ T cells in regulating the response of CD8+ ‘killer’ T cells, where we have found that the degree of inflammation associated with the priming stimulus will determine whether the CD4+ T cell response will help or hinder (regulate) the CD8+ T cells. This work has revealed unexpected roles for CD4+ T cells as well as for specific receptors that sense infectious pathogens, and will help to inform superior vaccination strategies for cancer and infectious disease. A 2nd line of research is directed towards the development of preclinical animal models of cancer immunotherapy, both in the context of adoptive immunotherapy as well as neoantigen-specific cancer vaccines. This work involves identifying the neoantigens expressed in murine and human tumors and optimizing methods for their specific targeting by various targeted vaccines or through adoptive cellular therapy (ACT) with neoantigen-specific T cells. Significant progress has been made in this area through our discovery of a process through which a patient’s tumor cells can be converted to cancer stem cells that retain expression of the neoantigens identified in the original cancer and which can form tumors in immunodeficient mice. Lastly, my laboratory is part of a clinical Head and Neck Cancer immunotherapy Program at UC San Diego’s Moores Cancer Center that is focused on treating patients through neoantigen-specific immunotherapy. Our work in this context is directed towards identifying neoantigens as well as the T cell receptors that recognize them through next-generation sequencing, cellular immunology, and single-cell genomics.