Heart Disease
Heart disease is the leading cause of death in the United States, according to the U.S. Centers for Disease Control and Prevention. Heart disease, also called cardiovascular disease, can describe several conditions, including coronary artery disease and aortic athlerosclerosis.
Atherosclerosis is the main underlying cause of most heart attacks. Patients with atherosclerosis have a build-up of deposits, or plaques, of cholesterol, calcium, and other substances in arteries. Over time, plaques limit blood flow, and, if they obstruct a vessel like the coronary artery, can trigger a heart attack. Artery blockage in other organs is also hazardous and can damage kidney, the extremities (in the case of peripheral artery disease), or the brain. Plaque rupture also increases the chance of stroke or blood clots in the brain.
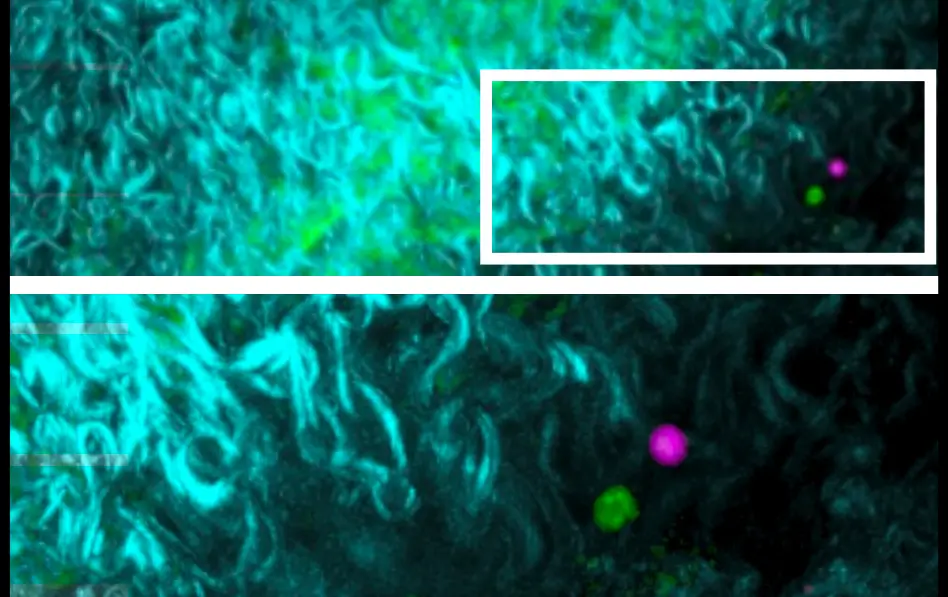
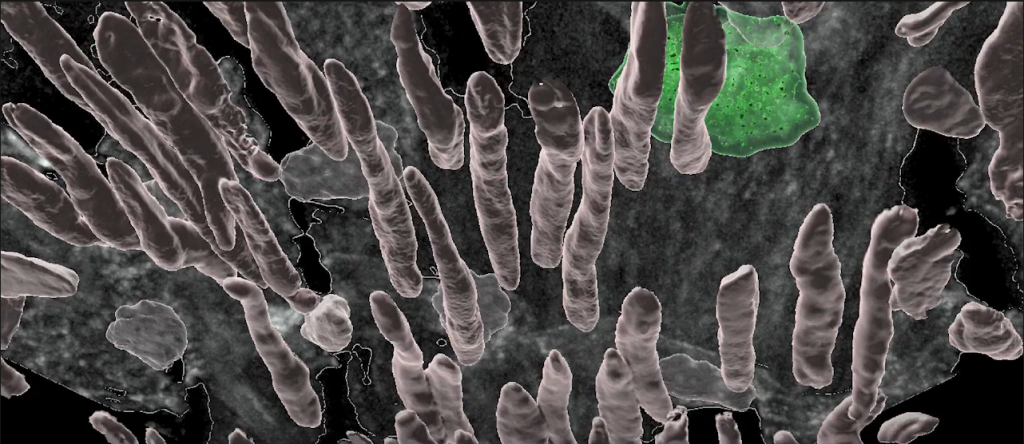
Scientists have found that immune cells and signaling molecules play a role in the development of several types of cardiovascular diseases. Immune cells can drive inflammation in the arteries and contribute to the buildup of damaging plaques in atherosclerosis.
Our Approach
LJI scientists are studying the roles of immune cells in the development of cardiovascular diseases such as atherosclerosis. Their work suggests new therapies might dial down vascular inflammation and help reduce arterial plaques.
As part of this effort to combat heart disease, LJI scientists are taking a closer look at how sex-based differences in the immune system affect cardiovascular health. This research may shed light on why more women than men die of heart disease each year.
LJI Associate Profess Sonia Sharma, Ph.D., is leading research into the roles of signaling molecules called bioactive lipids, show up in dramatically different numbers in men and women with inflammatory diseases. Her laboratory has found that these bioactive lipids could fuel inflammation and contribute to sex-based differences in how men and women develop heart disease, autoimmune vasculitis, and many other conditions.
Related News
- Research News
- Research News
- Research News
Research Projects
ATHEROSCLEROSIS Working in collaboration with LJI Professor Klaus Ley, the Sette Lab aims to help develop vaccines that target inflammatory factors as a means to reduce atherosclerotic plaque formation. The [...]