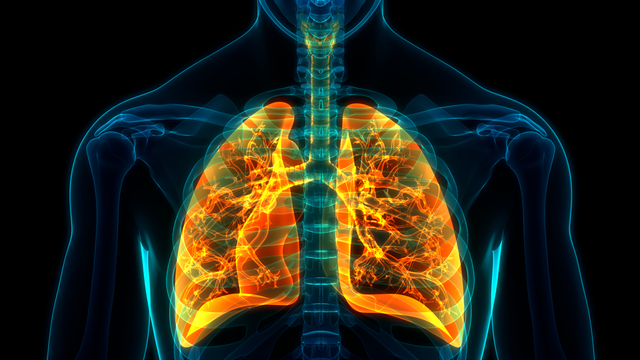
Allergies
Every year, 50 millions Americans experience a type of allergy. In fact, allergies are the sixth leading cause of chronic illness in the United States.
Allergies are caused by hypersensitivity to normally harmless molecules. When a person with an allergy encounters a particular protein or carbohydrate, they can experience an inflammatory immune response caused by antibodies known as IgE. When IgE molecules bind to mast cells, they trigger the release of histamine, which causes common allergy-related symptoms, such as coughing, runny nose (rhinitis) or itchy eyes. Histamine also dilates blood vessels, possibly to allow bulky white blood cells to race through the bloodstream in an allergic attack. Severe allergies can cause an acute “anaphylactic” allergic response, where blood pressure plummets to fatal levels.
There is a huge range of allergens, but the most common include pollen, nuts, milk, shellfish, latex and bee stings. As many as 10 percent of people are allergic to normally life-saving medications like penicillin. While some people “grow out” of childhood allergies, other allergies can affect ongoing quality of life. Physicians are also concerned that climate change may exacerbate plant-based allergens, as well as indoor mold growth and toxic air pollutants.
Conditions such as eczema, contact dermatitis, allergy-induced asthma and hay fever are all connected to allergy. While there are some therapies, such as allergic immunotherapies (“allergy shots”), these do not work for all patients.
Our Approach
Scientists at La Jolla Institute for Immunology (LJI) are studying the genetic causes and cellular response to allergens in an effort to make allergic immunotherapeutics more efficient and even tailored to specific patients.
LJI scientists are leaders in the area of allergy research. LJI Professor Emeritus Toshiaki Kawakami, M.D., Ph.D., devoted his career at LJI to studying allergic diseases at the basic as well as clinically relevant levels. His group revealed the role a secreted protein named histamine-releasing factor (HRF) as an essential pro-inflammatory effector in asthma and food allergy. The team generated inhibitors of HRF that can prevent these diseases in animals and worked to translate these inhibitors to human diseases. Dr. Kawakami also investigated signaling transduction of mast cell activation and developed better animal models of allergic diseases, including atopic dermatitis.
Today, LJI Professors Alessandro Sette, Dr. Biol. Sci., and Bjoern Peters, Ph.D., are working to identify the sites on allergen molecules that trigger an inflammatory immune response. They have been working on a variety of different allergens, including milk and peanut allergens, pollens, fungi, dust mites, mice and cockroach allergens. Apart from the experimental work they also lead a free, online resource called the Immune Epitope Database (IEDB), which collects and shares allergen data with scientists around the world.
Dr. Sette is also investigating cockroach allergy, which is a major contributor to asthma development in urban settings. As part of a large NIH-funded effort to develop immunotherapies for this allergy (the Inner City Asthma Consortium, or ICAC), the Sette lab efforts characterize T cell responses in allergic individuals before and after immunotherapy.
And researchers in Dr. Peters’s lab have analyzed immune cells in dozens of blood samples from allergy patients receiving allergy shots. Their work has shown that allergy immunotherapy shifts the balance between two types of immune cells: those that help churn out antibodies and those that knock down antibody production. As the latter gain the upper hand, the flood of allergy-associated IgE antibodies gradually decreases and patients’ symptoms start to improve. Currently, Dr. Peters is researching how these and other findings in inhaled allergens translate to food allergies, specifically those that affect children.
LJI Professor Pandurangan Vijayanand, M.D., Ph.D., focuses on using genomic and epigenomic tools to understand the molecular basis of asthma. His laboratory has developed techniques to study the molecular profiles of circulating and airway immune cells from patients with asthma and other diseases, using fewer cells than was possible previously. He aims to understand the epigenetic and genetic variations that contribute to a person’s susceptibility to diseases like asthma, lung cancer, and other pulmonary diseases.
Dr. Vijayanand and his colleagues recently found that a previously unknown population of T cells may help “dampen” the allergic response to house dust mites, a very common cause of allergies and asthma. This finding suggests that there may be a way to boost this T cell population to reduce responses to house dust mites and other allergens.
Learn More:
Related News
- Research News
Research Projects
Selected References Grégory Seumois, Ciro Ramírez-Suástegui, Benjamin J. Schmiedel, Shu Liang, Bjoern Peters, Alessandro Sette, Vijayanand P (2020). Single-cell transcriptomic analysis of allergen-specific T cells in allergy and asthma. [...]