Type 1 Diabetes
Type 1 diabetes is an autoimmune disease that affects nearly 1.6 million people in the United States. An autoimmune disease is a condition where the body’s own immune cells turn “self-reactive” and mistakenly attack healthy tissue, triggering inflammation and cell death.
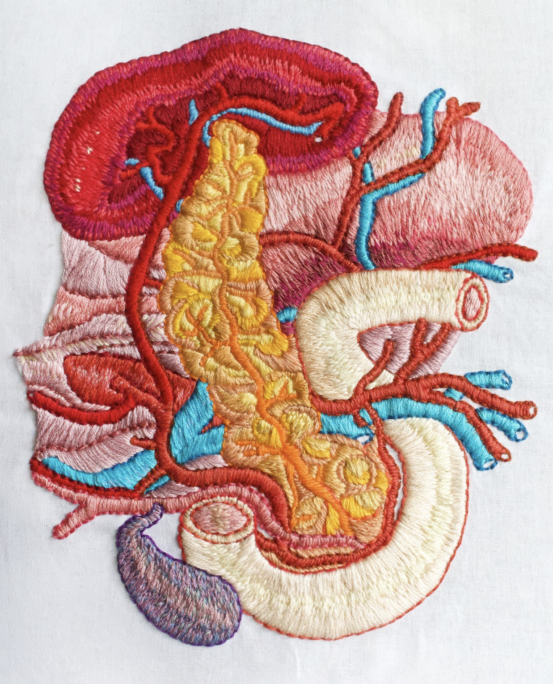
In patients with type 1 diabetes, self-reactive T cells kill beta cells in an organ called the pancreas, destroying the body’s ability to produce insulin. Without insulin, glucose cannot enter cells to produce energy. When people with type 1 diabetes experience a glucose build up in the blood, they can suffer from dehydration, weight loss, nerve and blood vessel damage, organ damage and life-threatening diabetic ketoacidosis.
Scientists are still investigating the root cause—or causes—of type 1 diabetes. Type 1 diabetes is not linked to a person’s diet or eating habits. Some people appear to be genetically predisposed to type 1 diabetes, but that does not guarantee they will develop the disease. There is no cure for type 1 diabetes; instead, people with the disease must carefully manage their health by monitoring their blood sugar and taking insulin shots.
Our Approach
Researchers at La Jolla Institute for Immunology (LJI) are exploring the hypothesis that there’s something in the pancreas driving its own destruction in type 1 diabetes. Scientists have found that beta cells draw harmful T cells to the pancreas prior to the onset of clinical signs of diabetes. This work has led LJI scientists to investigate the theory that there’s something wrong with the beta cells themselves.
One goal at LJI is to unravel the roles of different immune cells, cytokines, and other signaling molecules as type 1 diabetes progresses. LJI scientists analyze human pancreas tissues obtained from the Network for Pancreatic Organ Donors with Diabetes (nPOD), and also human isolated islets.
[Read more: Unmasking the pancreas]
By approaching type 1 diabetes from several angles, LJI scientists hope to unlock the root causes of many autoimmune diseases.
An infectious disease connection
LJI Instructors Mehdi Benkahla, Ph.D., and Estefania Quesada-Masachs, M.D., Ph.D., are investigating how predisposing genes and environmental factors, such as viral infections, may interact and trigger the disease. Several viruses, mainly from the Enterovirus genus have been considered as potential causal agents for type 1 diabetes. Their goals are to shed light on the mechanisms by which viruses trigger or exacerbate type 1 diabetes and to identify potential therapeutic targets to prevent or treat this devastating disease.
A nervous system connection
The pancreas is studded with islets, the cell clusters that house insulin-producing beta cells. In people with type 1 diabetes, the body’s own immune cells head for the islets and start attacking the beta cells. No one knows exactly what triggers this attack.
One clue may lie in the pattern of beta cell death. Many beta cells are killed off in big patches while other beta cells are mysteriously untouched. Something seems to be drawing immune cells to attack specific groups of beta cells while ignoring others. [Read more: Neuroimmunology]
In a 2020 Science Advances study, LJI researchers reported that the nervous system may be driving this patchy cell die-off. Their new findings in a mouse model suggest that blocking nerve signals to the pancreas could stop patients from ever developing type 1 diabetes.
Examining T cell behavior
It’s long been thought that having “autoreactive” T cells in the pancreas was a sure sign of type 1 diabetes. In 2020, an LJI-led study showed that even healthy people have these cells lurking in the pancreas—in surprisingly high numbers. It appears that high numbers of these T cells in the pancreas are the default—whether you have type 1 diabetes or not. In people with type 1 diabetes, tissue samples showed autoreactive T cells very close to—and even infiltrating—the islets.
These results add evidence for the theory that type 1 diabetes is not caused by malfunctioning T cells attacking beta cells. Instead, the body is already making these T cells and something in the pancreas is triggering the attack. The findings also suggest that an effective type 1 diabetes therapy would need to be local to the pancreas. [Read more: A cold case no longer]
Going forward, the researchers plan to take a closer look at how the preproinsulin-specific T cells behave. The team also hopes to investigate other proteins in islets that might attract T cell attacks.
Learn more:
Related News
- Institute News
- Immune Matters
- Research News
Research Projects
Team: Estefania Quesada-Masachs, Sakthi Rajendran Type 1 diabetes (T1D) is an autoimmune disease in which insulin-producing β cells are attacked by the immune system. Earlier work suggests that β cells [...]
Team: Sakthi Rajendran, Estefania Quesada-Masachs and Mehdi Benkahla IL-17 is a pro-inflammatory cytokine important in shaping host immune responses against pathogens. IL-17 is also attributed to cause tissue damage and [...]
Team: Sakthi Rajendran, Estefania Quesada-Masachs, Christine Bender Type 1 diabetes (T1D) is an autoimmune disease caused by the destruction of insulin-producing β cells by the immune system. However, the process [...]